Five reasons why you should not use oxyhydrogen or browns gas!
This blog post is dedicated to showing you the truth about using oxyhydrogen versus molecular hydrogen alone. It is staggeringly more dangerous to use oxyhydrogen. Read this blog post and find out why.
Jim Mitchell founder molecularhydrogenbubbles.com
4/8/20264 min read


5 Reasons You Should NOT Use Oxyhydrogen Instead of Molecular Hydrogen Alone.
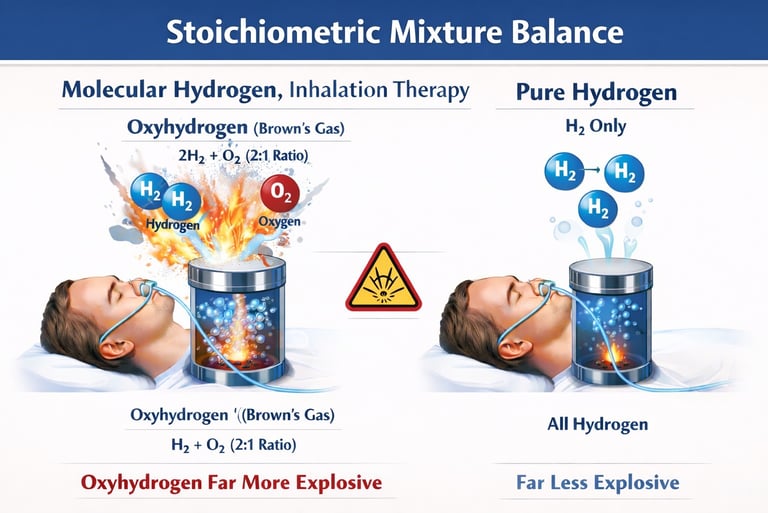
1.Molecular hydrogen therapy is based on a straightforward principle: deliver hydrogen safely within a therapeutic, noncombustible concentration range, typically between 2-4%. However, some devices on the market generate or deliver oxyhydrogen a mixed gas containing hydrogen (H₂) and oxygen (O₂), commonly referred to as (HHO) or Brown’s gas. This is not a minor variation of molecular hydrogen therapy, it is an entirely different substance with fundamentally different chemical properties, physical behavior, and safety considerations. Below are five critical reasons why pure molecular hydrogen delivery is preferable to oxyhydrogen gas. Oxyhydrogen Is premixed Fuel plus Oxidizer (That’s What Explosives Are). An explosion requires three things, fuel, oxidizer, ignition oxyhydrogen already contains two ingredients except the spark. Hydrogen = fuel, oxygen = oxidizer, add static electricity = ignition. This is why oxyhydrogen has historically been called “knallgas” (detonating gas). In contrast pure hydrogen alone is NOT explosive until it mixes with air that difference is everything.
2.The Explosion Range Is Enormous (4%-95%).
When hydrogen is mixed with oxygen, the result is an extremely broad explosive range. Hydrogen in air becomes flammable between approximately 4% and 75%, but hydrogen combined directly with oxygen can ignite or explode from about 4% all the way up to 95%. In practical terms, that means nearly any concentration of oxyhydrogen can be hazardous. This is completely different from therapeutic molecular hydrogen applications, which are intentionally kept below the combustible threshold typically in the 2%-4% range considered the therapeutic and noncombustible safety zone.
3. Peak Detonation Ratio.
Oxyhydrogen systems generate gas at or very near a 2:1 hydrogen-to-oxygen ratio (approximately 66% H2 to 33% O2), which represents the most explosive mixture possible for these elements. Scientific literature has long recognized this specific ratio as highly volatile, noting its capacity for violent detonation and what researchers describe as a “ferocious detonating sound.” Consequently, these systems are not producing hydrogen within a safe, diluted therapeutic range instead, they are generating a gas mixture at the chemically optimal ratio for rapid combustion and explosion.
4. Ignition Energy Is Extremely Low (Even Static Can Trigger It)! Hydrogen especially when premixed with oxygen—requires an extraordinarily small amount of energy to ignite. The minimum ignition energy is approximately:~0.007 millijoules For perspective, that is far below the energy produced by a tiny static electricity discharge. That means seemingly ordinary events could theoretically ignite the mixture, including: Plugging in a device Synthetic clothing generating static Dry indoor air A minor electrical spark In other words, oxyhydrogen mixtures can be ignited by energy sources so small they are often unnoticed.
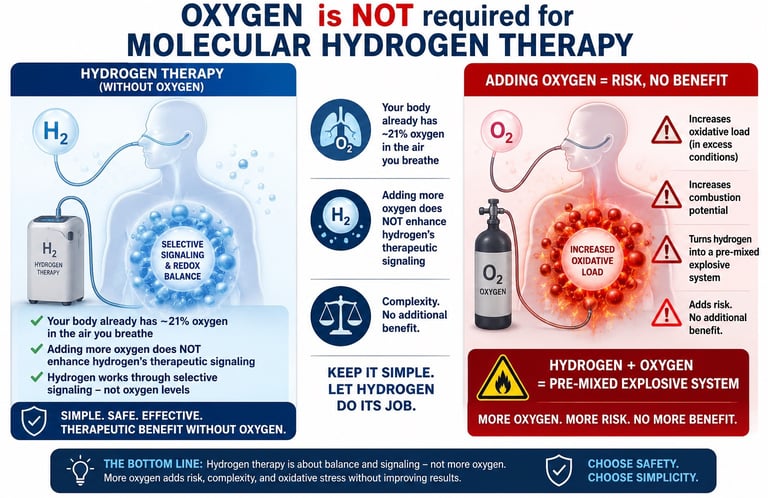
5.Here’s the part most people misunderstand Oxygen is NOT required for molecular hydrogen therapy. In fact: Your body already has ~21% oxygen in the air you breathe Adding more oxygen does not enhance hydrogen’s therapeutic signaling However, oxygen does Increase oxidative load (in excess conditions) increase combustion potential turn hydrogen into a pre-mixed explosive system so you’re adding risk Complexity no additional benefit.
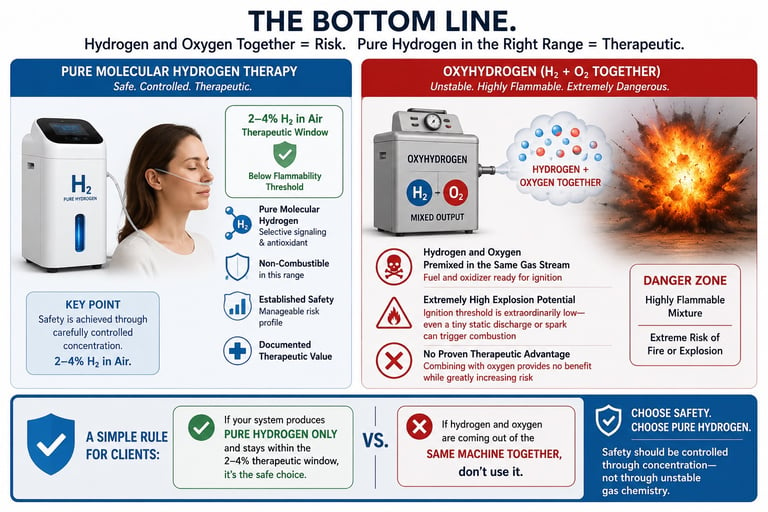
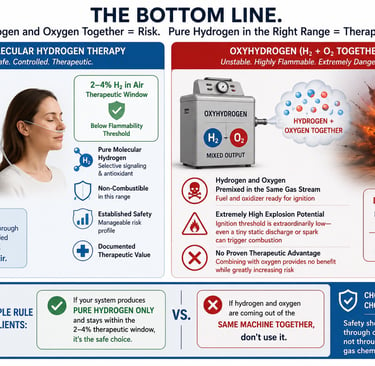
The Bottom Line.
Pure molecular hydrogen and oxyhydrogen are not the same thing. Therapeutic molecular hydrogen is delivered within a carefully controlled, non-combustible concentration range typically 2–4% hydrogen in air. In this range, the gas remains below the flammability threshold and maintains a manageable safety profile. Oxyhydrogen is fundamentally different. It combines hydrogen and oxygen together in the same gas stream, creating a highly unstable mixture with extreme explosion potential. Unlike pure hydrogen therapy, the fuel and oxidizer are already premixed and ready for ignition. The ignition threshold is extraordinarily low, meaning even a tiny static discharge or minor spark could theoretically trigger combustion. Molecular hydrogen has documented therapeutic value as a selective signaling and antioxidant molecule. Adding oxygen into the same gas stream provides no proven therapeutic advantage, while substantially increasing risk. A simple rule for clients: If hydrogen and oxygen are coming out of the same machine together, don’t use it. Instead, use systems that produce pure hydrogen output only, and stay within the established 2-4% therapeutic window. Safety should be controlled through concentration not through unstable gas chemistry.
Before choosing any hydrogen therapy device, understand exactly what gas the system is producing. Safety in molecular hydrogen therapy comes from controlled concentration not from mixing unstable gases together. If a machine is generating hydrogen and oxygen in the same stream, you are no longer dealing with simple molecular hydrogen therapy you are dealing with a highly reactive fuel-and-oxidizer mixture. Choose systems designed to deliver pure molecular hydrogen only, within the established 2–4% therapeutic range. Ask manufacturers direct questions. Request gas composition data. Prioritize safety, transparency, and evidence-based design over marketing claims. When it comes to molecular hydrogen therapy, simpler chemistry is safer chemistry.
Written by Jimmitchell founder
molecular hydrogenbubblesare.com